Referências:
-
1.
Bula profissional de Ofev® (esilato de nintedanibe). Versão aprovada pela ANVISA em 22Jun2020. pc-br-107012 bula profissional de ofev 2022.pdf (boehringer-ingelheim.com)
-
2.
Hilberg F, Roth GJ, Krssak M, et al. BIBF 1120: Triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res. 2008;68(12):4774-4782.
-
3.
Wollin L, Maillet I, Quesniaux V, Holweg A, Ryffel B. Antifibrotic and anti-inflammatory activity of the tyrosine kinase inhibitor nintedanib in experimental models of lung fibrosis. J Pharmacol Exp Ther. 2014;349(2):209-220, doi:10,1124/ jpet.113.208223.
-
4.
Wollin L, Wex E, Pautsch A, et al. Mode of action of nintedanib in the treatment of idiopathic pulmonary fibrosis. Eur Respir J. 2015;45(5): 1434-1445. doi:10,1183/09031936.00174914.
-
5.
Wollin L, Distler JHW, Denton CP, Gahlemann M. Rationale for the evaluation of nintedanib as a treatment for systemic sclerosis–associated interstitial lung disease. J Scleroderma Relat Disord. 2019;4(3):212-218.
-
6.
Wollin L, Distler JHW, Redente EF, et al. Potential of nintedanib in treatment of progressive fibrosing interstitial lung diseases. Eur Respir J. 2019;54:1900161.
-
7.
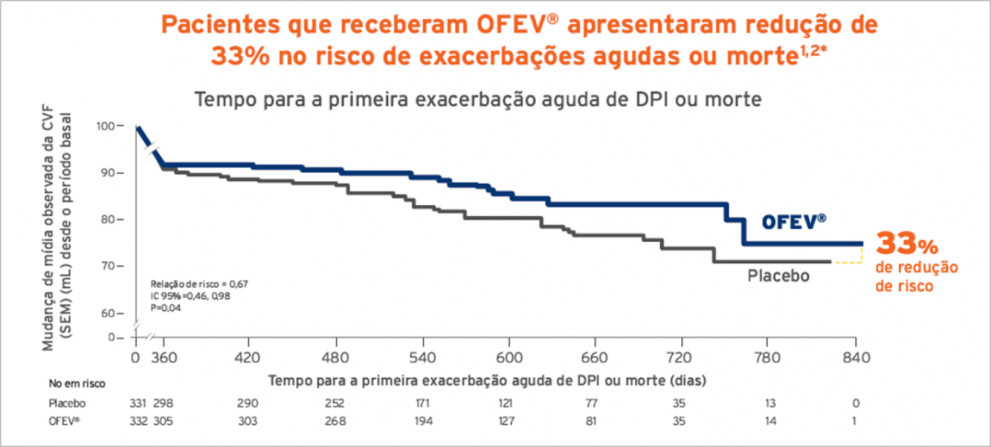
Flaherty KR, Wells AU, Cottin V, et al. Nintedanib in progressive fibrosing interstitial lung diseases. N Engl J Med. 2019;381(18):1718-1727. doi: 10,1056/NEJMoa1908681.
-
8.
Richeldi L, Du bois RM, Raghu G, et al. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22):2071-82.
-
9.
Distler O, Highland KB, Gahlemann M, et al. Nintedanib for Systemic Sclerosis-Associated Interstitial Lung Disease. N Engl J Med. 2019; Jun 27;380(26):2518-2528.
-
10.
Flaherty KR, Wells AU, Cottin V, et al. Nintedanib in Progressive Fibrosing Interstitial Lung Diseases. N Engl J Med. 2019;381:1718-27.
-
11.
Richeldi L, Costabel U, Selman M, et al. Efficacy of a tyrosine kinase inhibitor in idiopathic pulmonary fibrosis. N Engl J Med. 2011;365(12):1079-87.
-
12.
Crestani, B, Huggins, JT, Kaye, M. Long-term safety and tolerability of nintedanib in patients with idiopathic pulmonary fibrosis: results from the open-label extension study, INPULSIS-ON. Lancet Respir Med 2019:7:60-68.
-
13.
Distler O, Highland KB, Gahlemann M, et al. Nintedanib for Systemic Sclerosis-Associated Interstitial Lung Disease. N Engl J Med. 2019; Jun 27;380(26):2518-2528. doi: 10.1056/NEJMoa1903076. Epub 2019 May 20.
-
14.
Brown KK et al. Does HRCT pattern influence the effect of nintedanib in patients with progressive fibrosing interstitial lung diseases (ILDs)? Poster developed for the American Thoracic Society International Conference, 2020.
-
15.
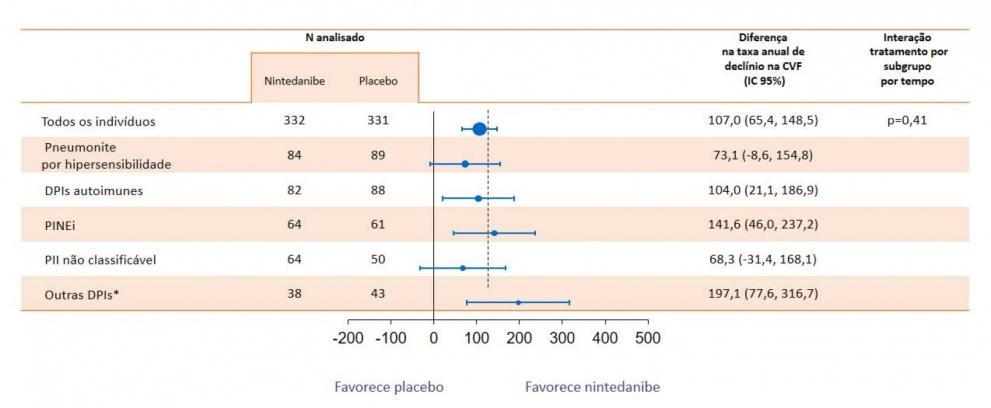
Wells AU, Flaherty KR, Brown KK, et al. Nintedanib in patients with progressive fibrosing interstitial lung diseases-subgroup analyses by interstitial lung disease diagnosis in the INBUILD trial: a randomised, double-blind, placebo-controlled, parallelgroup trial. Lancet Respir Med. 2020; doi: 10.1016/S2213-2600(20)30036-9.
-
16.
Flaherty KR, Wells AU, Cottin V, et al. European Respiratory Journal Sep 2019, 54 (suppl 63) RCT1881; DOI: 10.1183/13993003.congress-2019.RCT1881